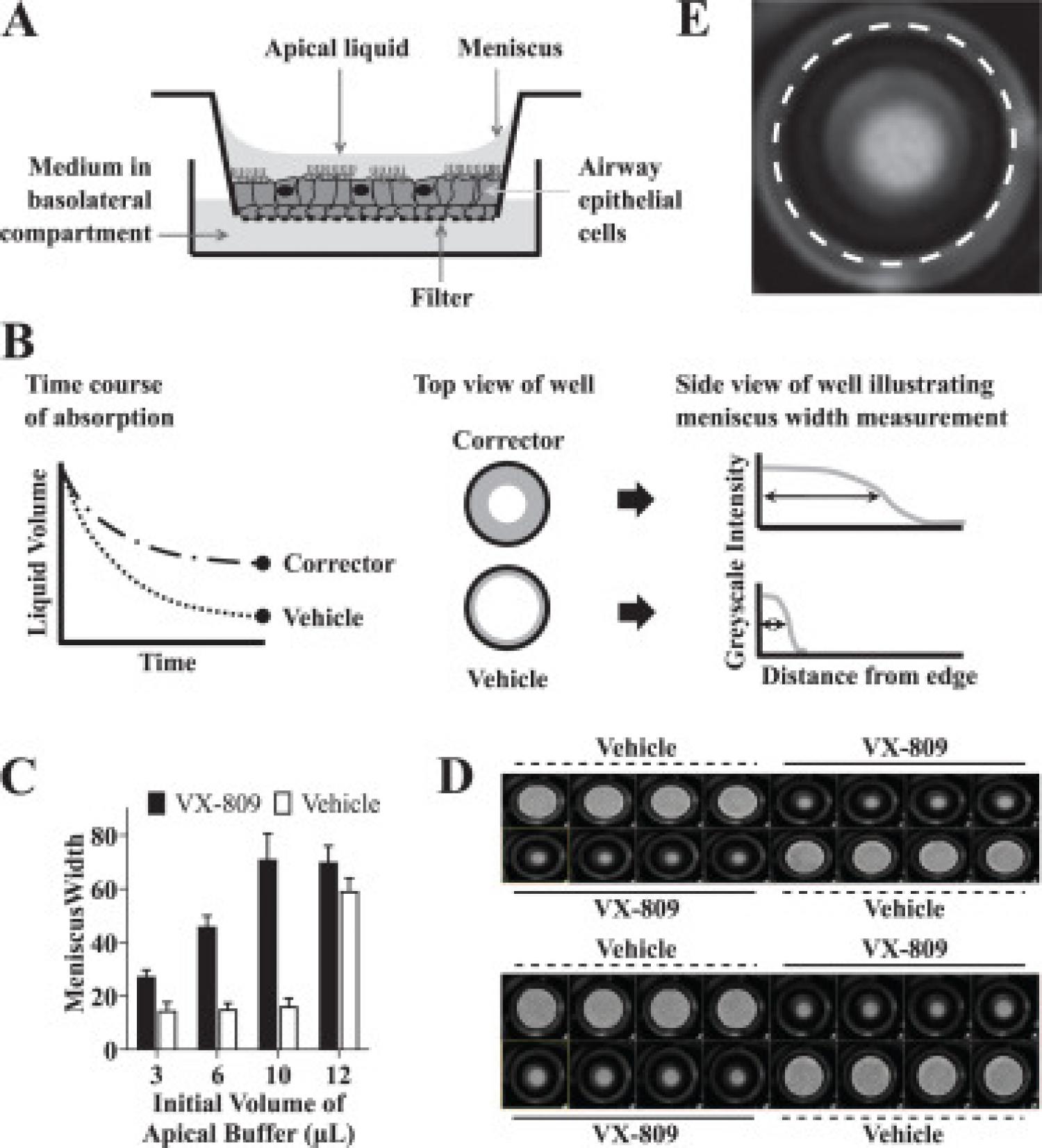
High-throughput screening for drug discovery is increasingly utilizing cellular systems of high physiological relevance, such as patient primary cells and organoid cultures. We used 3D-cultured cystic fibrosis patient bronchial epithelial cells to screen for new small-molecule correctors of the disease-causing F508del mutation in CFTR. Impaired mucociliary clearance due to insufficient airway hydration is a hallmark of cystic fibrosis and we used a simple measure of surface liquid levels to quantify F508del CFTR correction in cultured bronchial epithelial cells. Two robust assay formats were configured and used to screen more than 100,000 compounds as mixtures or individual compounds in 96-well format. The corrector discovery success rate, as measured by the number of hits confirmed by an electrophysiology assay on patient primary bronchial epithelial cells, was superior to screens in cell lines expressing recombinant F508del CFTR. Several novel corrector scaffolds were discovered that when combined with the clinical corrector VX-809 delivered combination responses greater than double that of VX-809 alone. This work exemplifies the advantages of a disease-relevant readout and 3D-cultured patient primary cells for the discovery of new drug candidates.